Research & Development
Our Approach to Region-Selective Drug Discovery
Targeted CNS therapeutics designed to act where neural activity goes wrong—not across the whole brain.
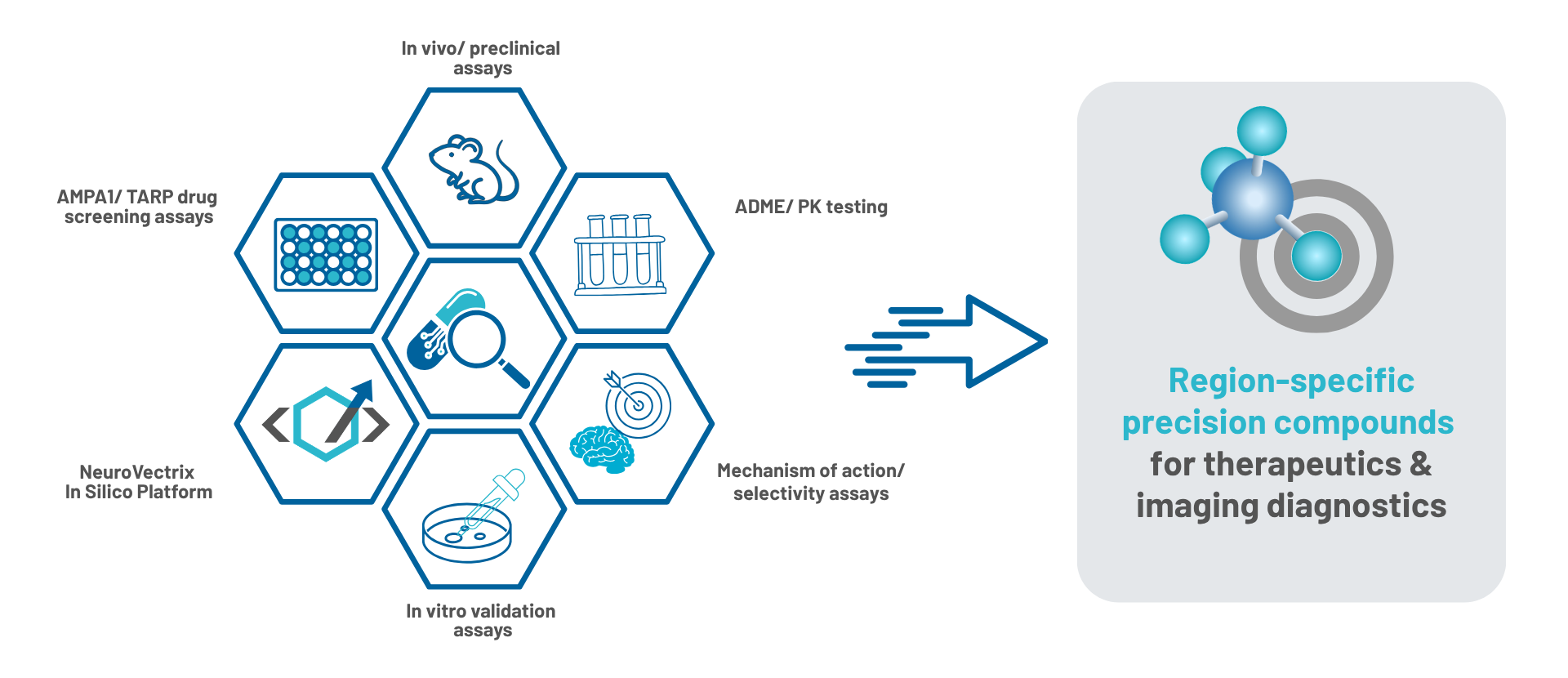

We are advancing a pipeline of precision CNS therapeutics enabled by our region and circuit-targeting drug discovery platform. The platform integrates proprietary technologies into a unified engine that combines real-world biological systems with AI and machine learning to continuously learn from experimental data—improving predictions and accelerating compound optimization. In vitro and in vivo assays enable high-fidelity screening in physiologically relevant contexts, while our computational platform unifies generative chemistry, SAR analysis, and predictive modeling. These capabilities are orchestrated by our Discovery Autopilot agent, which autonomously designs and evaluates compounds through end-to-end computational cycles, seamlessly integrating with downstream synthesis and experimental testing.
At its core, our approach bridges wet lab and in silico discovery in a closed-loop system, where autonomous computational workflows and human expertise work in tandem to design and validate compounds.

Targeting Glutamate Receptors with Circuit-Level Precision
Ionotropic glutamate receptors, including AMPA receptors, are central to excitatory neurotransmission and the regulation of neural circuit activity. When their function becomes dysregulated, it can disrupt circuit balance and drive a wide range of neurological and neuropsychiatric disorders. However, existing approaches to modulating glutamate signaling often lack regional specificity, leading to broad effects across the brain and limiting both efficacy and tolerability.
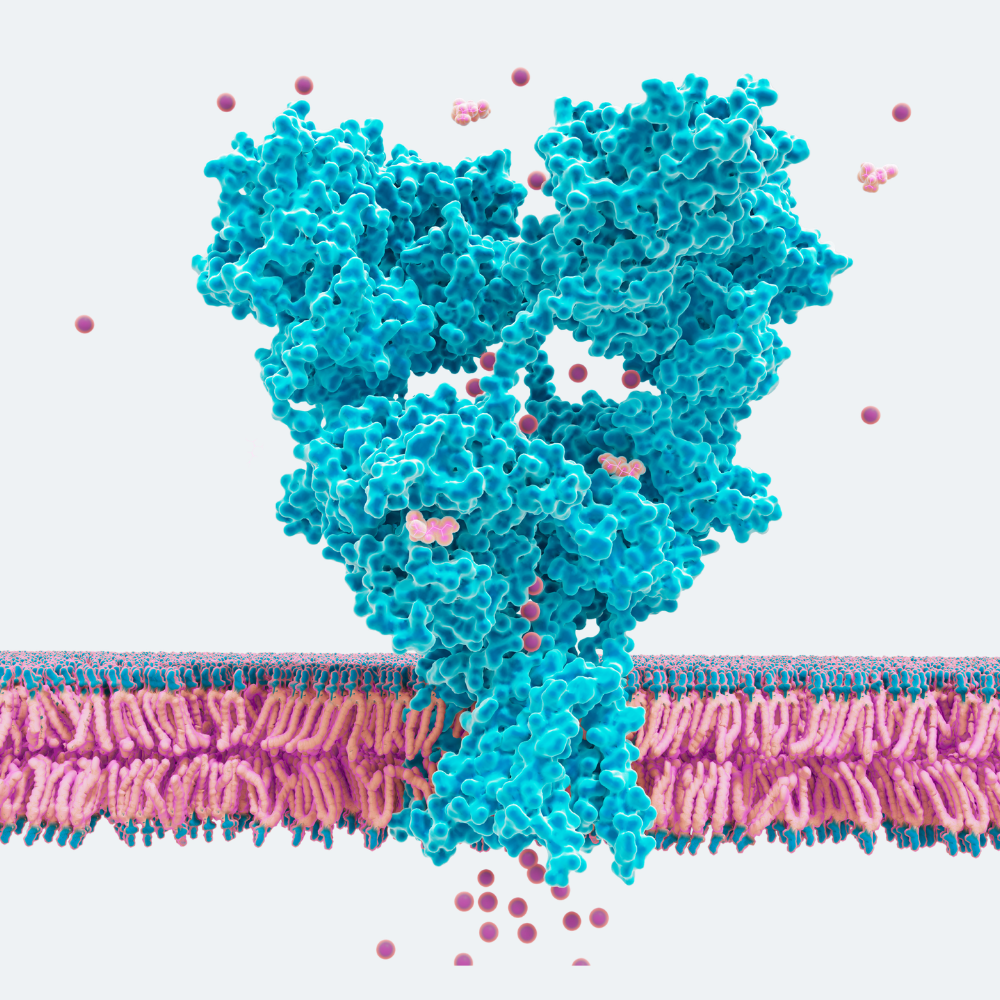
To address this challenge, we leverage the unique biology of transmembrane AMPA receptor regulatory proteins (TARPs). TARPs co-localize with AMPA receptors and play a critical role in governing their function, trafficking, and localization. Importantly, their distinct expression patterns across brain regions and neural circuits provide a powerful mechanism for achieving spatial precision in drug design.
By targeting TARP-associated AMPA receptor complexes, we direct therapeutic activity to disease-relevant circuits and unlock novel binding sites inaccessible to conventional approaches. This enables a new class of precision neuromedicines designed to modulate dysfunctional circuits and restore healthy activity and plasticity—potentially improving efficacy while reducing off-target effects.

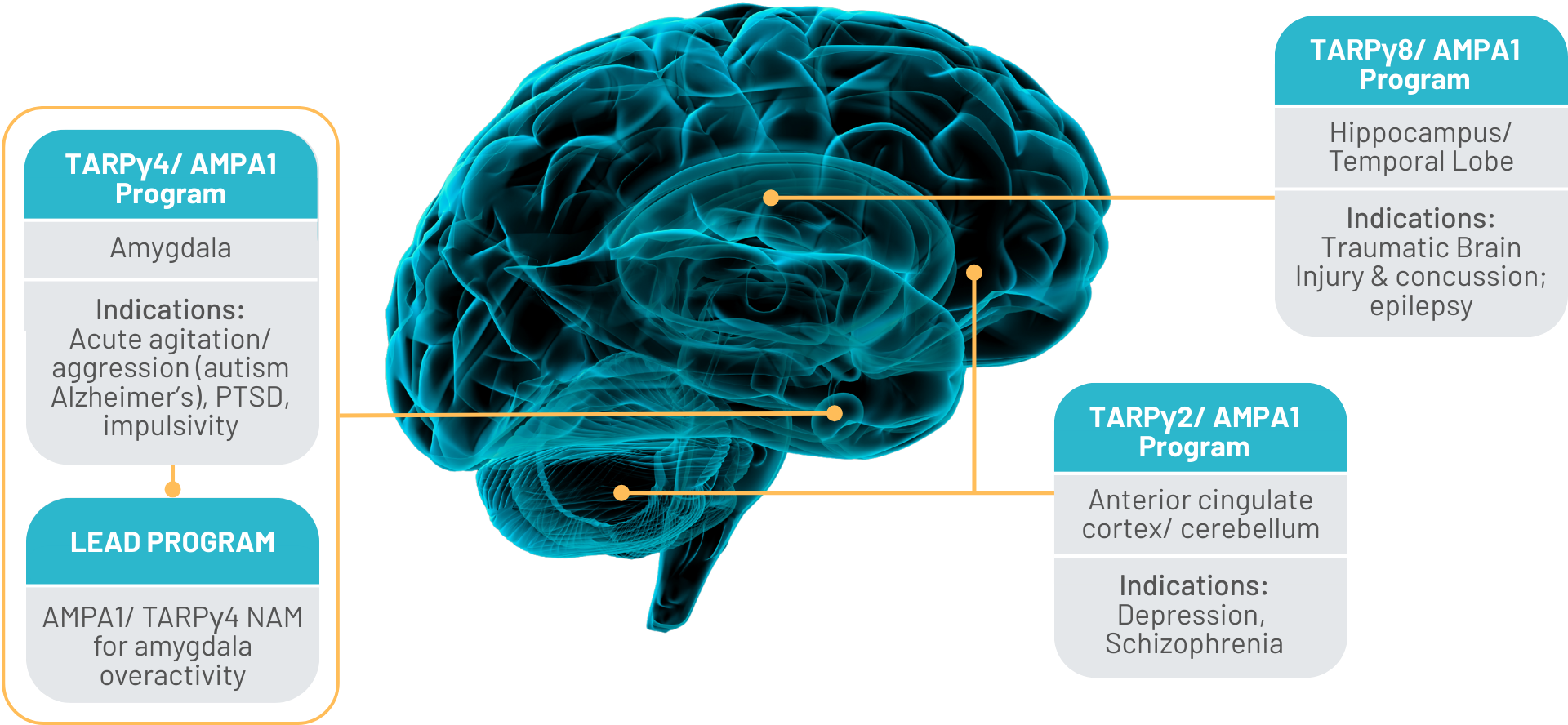
Programs
We are building a pipeline of first-in-class, regionally selective AMPA receptor modulators powered by our TARP-based platform.
Our programs are designed to address disorders driven by circuit-level dysfunction, where existing therapies fall short due to lack of specificity. By targeting TARP-defined receptor populations, we aim to deliver differentiated therapeutics with improved efficacy and reduced side effects.
Multiple programs are currently in discovery, establishing a scalable path toward clinical development. Our lead program focuses on selective modulation of GluA1-containing AMPA receptors associated with TARPγ4 for amygdalar overactivity.

